
Making cold process soap successfully requires precise measurements of lye and oil. If too much lye is used the soap will be lye heavy, which can irritate or even burn the skin. Lye heavy soap will have a higher pH than perfectly balanced cold process soap. If you are worried that your soap is lye heavy, there are several techniques to test the pH and discover if it is safe to use on the skin. One of which includes using something you may already have in your fridge…red cabbage!
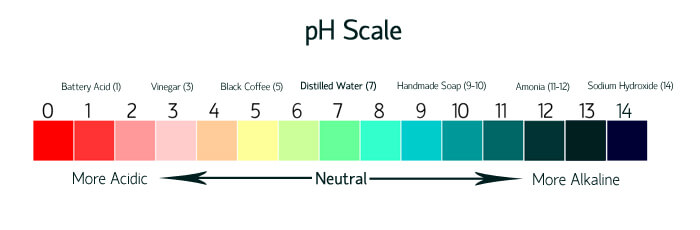
What is pH and what does it have to do with soap? The pH scale measures how acidic or basic a substance is. The scale ranges from 0 to 14. A pH level of 7 is considered neutral. A pH higher than 7 is considered basic or alkaline, and pH level of below 7 is acidic. Cold process soap is naturally alkaline with a pH of around 9-10. This pH helps to gently clean the skin.
 Sodium hydroxide lye has a pH of about 14, putting it at the very top of the pH scale. When too much lye is used in the soap, the pH level will rise from the normal level of 9-10, and can become closer to 11-14. Why is this? Simply put, when too much lye is used there is not enough oil to turn it into soap. This lye is “free-floating” in the soap which increases the pH level. Soap with high pH will irritate or burn the skin and should not be used.
Sodium hydroxide lye has a pH of about 14, putting it at the very top of the pH scale. When too much lye is used in the soap, the pH level will rise from the normal level of 9-10, and can become closer to 11-14. Why is this? Simply put, when too much lye is used there is not enough oil to turn it into soap. This lye is “free-floating” in the soap which increases the pH level. Soap with high pH will irritate or burn the skin and should not be used.
If you do end up with lye heavy soap, one way to recycle it is to turn it into laundry soap. Check out this blog post for three different recipes for laundry soap. If your soap was made recently, you can add missing essential fatty acids (oils you shorted in the original recipe) and use the Hot Process Hero method to “cook” the soap to the balanced pH level. This method will only work within 24 hours of making your soap. Hot Process Hero also a great method to fix batches that have separated or seized.
There are a few different ways to test for lye heavy soap. One of the most common is to use pH strips. To test cold process soap, simply wet the soap with distilled water and rub your hands on the soap to form bubbles. Place the pH strip on the wet soap to get a pH reading. If you don’t have pH strips on hand, the “zap test” is another method to test pH. Place the bar of soap onto the tip of your tongue. If you feel a slight zap feeling (similar to licking a battery), this means your soap is lye heavy. Please note that pH strips do not work if you’re using tap water; only use distilled water for this methods.
The old-timers tongue test is an effective way to test for excess lye. If you lick your soap and it ‘zaps’, you know it’s lye heavy. If your soap initially zaps after taking it out of the mold, don’t panic. Wait the full 4 to 6 week cure time before retesting. Hopefully in 4 to 6 weeks, your soap will taste like… soap! Often fresh soap gives a little zing as it finishes its curing and drying process.
Don’t have pH strips, and don’t want to lick soap? Red cabbage (aka: purple cabbage) is an effective pH indicator. When the juice of red cabbage is placed onto soap, it changes color depending on the pH level. To prepare the juice, roughly chop a 3-4 leaves of the cabbage. Place the cabbage in a large bowl and add about 1 cup of distilled water. Distilled water is extremely important – if the water is not distilled, the test will not work. Tap water can have microbes or bits of metal in it, which will affect the test results.
 Use a stick blender to puree the cabbage. You can also place the cabbage into a blender, or use a juicer if you have one. Once blended, strain out the cabbage pieces and discard.
Use a stick blender to puree the cabbage. You can also place the cabbage into a blender, or use a juicer if you have one. Once blended, strain out the cabbage pieces and discard.

 Once you have the cabbage liquid, it’s ready to use! Using a dropper or spoon, place a small amount of the juice directly onto your soap. Within seconds, the purple juice will begin to change color. Red cabbage contains a pigment molecule called flavin that changes color depending on the pH level of various substances. If the pH is a safe level for soap (8-10) the purple liquid will quickly change from purple to blue.
Once you have the cabbage liquid, it’s ready to use! Using a dropper or spoon, place a small amount of the juice directly onto your soap. Within seconds, the purple juice will begin to change color. Red cabbage contains a pigment molecule called flavin that changes color depending on the pH level of various substances. If the pH is a safe level for soap (8-10) the purple liquid will quickly change from purple to blue.
 Below is a soap made just a few days before testing (left), while the other soap has cured for about three weeks. You can see that neither soaps are lye heavy, as the color has changed from purple to blue. As you can see, this test works well for freshly made soap, as well as soap that has cured for several weeks.
Below is a soap made just a few days before testing (left), while the other soap has cured for about three weeks. You can see that neither soaps are lye heavy, as the color has changed from purple to blue. As you can see, this test works well for freshly made soap, as well as soap that has cured for several weeks.
 If soap is lye heavy, the purple cabbage juice will turn from purple to green or yellow. The soap below contains 1% excess lye. Notice the green color. Over an 8 hour period, this green tone changed from the deep green shown here to a lighter yellow.
If soap is lye heavy, the purple cabbage juice will turn from purple to green or yellow. The soap below contains 1% excess lye. Notice the green color. Over an 8 hour period, this green tone changed from the deep green shown here to a lighter yellow.
 Below is the same lye heavy soap as above; notice how the green color has become a lighter hue. On the right, is a soap made with only 1% super fat. The red cabbage juice quickly turned from a deep purple color to a blue.
Below is the same lye heavy soap as above; notice how the green color has become a lighter hue. On the right, is a soap made with only 1% super fat. The red cabbage juice quickly turned from a deep purple color to a blue.

This method of testing the pH level of soap is a lot of fun, and would make an good learning-activity project if you have kiddos. (Side note: Did you know I wrote an entire book of activities for kids around soap? It’s designed to be a supplement to a home-schooling program. Check it out here). Once the cabbage juice is made, it can be left in a airtight container in the fridge for about a week. Have you ever ended up with a lye heavy batch of soap? It happens to the best of us. Luckily, there are plenty of ways to test before using it on the skin.

Hi there! I need some help. I made a batch of soap and subbed orange juice for water. I have been doing some research and found that if you use ingredient woth a higher sugar content you can keep the soap from exploding during gel phase if you put it in the freezer. So I made a 2 pound batch and put one pound in the freezer and insulated and gelled the other pound. The freezer soap I transfered to the fridge and 48 hours later I went to cut the both. The non gelled soap is a gorgeous creamy white and the gelled is the usual kind brown color I’ve been getting. So I went to cut them both and the gelled soap cut nicely. The ungelled fridge soap is super crumbly and falling apart. What did I do wrong? Thank you!
Hi Julia!
Hmm, I’m not sure why that happened! Typically, if soap gets too hot it can be brittle or crumbly. The soap that was kept cooler should be softer. Does it feel soft to the touch, or are the pieces harder? Also, what oils did you use in your recipe? Let me know and I’ll help you troubleshoot. 🙂
-Kelsey with Bramble Berry
Hi I love all about you create here. Btw I have make my our cold process lavender soap and I check PH level only stay at PH 7. Exit that soap to natural to use n don’t have any benefit??
Thanks a lot if u can reply my msg 🙂
Thank you so much Amy, glad you love the blog! That soap should still cleanse and moisturize the skin. The pH of 7 means it will be a bit more gentle than one with 9-10. Did you add any citric acid to the mix? That usually drops the pH level. 🙂
-Kelsey with Bramble Berry
Hello greetings!!! I’m new jaboneando and have problems with ph, my ph is at 10, is that correct? I can achieve lower the pH in cold process with citric acid, if positive as I use it and in what quantity?
very nice thanks.
A pH of 10 is just right! Soap pH is around 9-10. If you like, you can lower the pH with citric acid. That makes it a more neutral 7. Learn how in this Sudsy Shampoo Bar Cold Process Tutorial: https://soapqueen.com/bath-and-body-tutorials/cold-process-soap/sudsy-shampoo-bars/
-Kelsey with Bramble Berry
I don’t know if someone asked already but would this work with Koh paste. To test if it’s done?
Yes, the cabbage liquid should work for liquid soap paste as well! You can also use pH test strips. 🙂
pH Test Strips: https://www.brambleberry.com/pH-Strips-1-pack-of-50-P4434.aspx
-Kelsey with Bramble Berry
Hi,
I have recently attempted to make soap for the first time and it took about 2 days afterwards to achieve trace. I made a Castile soap with Spring Water/Extra Virgin Olive Oil/Organic Almond Oil and Organic Rose essential oil
My soaps have now finally began to harden and have been removed from their moulds.
I have now tested the PH level via both tongue and 2 x PH strips, their was a very mild tingling sensation with the tongue test and the strips indicated around the 12 mark.
I have also rubbed a tiny amount of the soap onto my hands and washed them and had no ill affects?
My question is this – during the curing period and the weeks in which they will harden, will the PH level adjust itself with time i.e. soften and reduce to a more useable/safer level of around 8-10?
If it is that the PH level will remain above 10, then is there anything else I can use these for?
Any help or advice would be greatly appreciated.
Kind regards
Terry
Hi Terry!
How long has it been since you made the soap? We usually recommend testing it after 5 days. Most of the saponification happens during those first 2 days, then the pH level should drop. If it’s still 12 pH, we don’t recommend using it on the skin because it can be irritating. You can use it for laundry soap though! Learn more about that here: https://soapqueen.com/bath-and-body-tutorials/tips-and-tricks/troubleshooting-lye-heavy-soap/
Typically, the soap should trace in about 1 minute! What are you using to blend the soap? Let me know and I’ll help you troubleshoot. 🙂
-Kelsey with Bramble Berry
Hi Kelsey,
Thank you so much for the reply.
I made the soap on the 10th June and having rechecked the PH yesterday (18th), the test strip appears to indicate that it has dropped somewhat to around the 10 mark. I tested it twice with the same result. I also did another tongue test and the tingling experienced the first time has also subsided to extremely mild. Additionally, I used a broken piece of the soap and washed my hands with no apparent ill affects except to say the skin felt soft and smooth. I was following a recipe from a book and followed everything spot on except that I substituted pam Oil for an equal amount of Almond Oil and being Vegan, I omitted 59g of beeswax that had been part of this recipe. I also didn’t adjust the Lye level accordingly or use another ingredient. Being a novice soap maker and believing that 59g is not a huge amount in terms of soap making, then I didn’t think it would matter too much? I did speak to the author about it but although she replied to my message, she didn’t appear to be that interested except to say – read the page on Saponification?
Please excuse my ignorance but I want sure what you are referring to with regards to what I used to blend the soap?
I made a Castile soap and the full ingredients I used were as follows – Spring Water/680g Extra Virgin Olive Oil/227g Organic Almond Oil and approx. 1 tsp. Organic Rose essential oil.
Any further advice would be greatly appreciated.
Kind regards
Terry
That’s awesome, it sounds like the soap is the correct pH level and will feel great on the skin! 🙂
If you make any substitutions or leave an ingredient out, we recommend running the recipe through a Lye Calculator again. Every oil has a slightly different lye amount, so the calculations may be a bit different. You can find our Lye Calculator here: http://www.brambleberry.com/Pages/Lye-Calculator.aspx
This post has more information on substituting oil in cold process recipes: https://soapqueen.com/bath-and-body-tutorials/tips-and-tricks/how-to-substitute-oil-in-cold-process-recipes/
Also, a stick blender is a tool used to emulsify soap! It’s also called an immersion blender. It emulsifies the oil and lye really quickly. You can find them online, at a kitchen supply store, a department store like Target or at BrambleBerry.com. 🙂
White Stick Blender: https://www.brambleberry.com/White-Stick-Blender-Kitchenaid-P5604.aspx
-Kelsey with Bramble Berry
hola me gustaria que alguien me comentara si el acido citrico lo puedo usar para bajar el ph en proceso en frio.
A pH of 10 is just right! Soap pH is around 9-10. If you like, you can lower the pH with citric acid. That makes it a more neutral 7. Learn how in this Sudsy Shampoo Bar Cold Process Tutorial: https://soapqueen.com/bath-and-body-tutorials/cold-process-soap/sudsy-shampoo-bars/
-Kelsey with Bramble Berry
Oh my I tried the cabbage test but I really cannot tell if blue or green!!! I would say sort of blueish green! My ph strips seem to indicate 10 but there is no zap! I super fatter 4% but had accelerated trace and ended up having to cook in crockpot to be sure everything blended. It went to gel in crock pot SUPER fast. I am going to give it a day or two then just try it on my skin. Any advice appreciated!!
Hi Sherry!
If that soap is at 10 pH, it’s absolutely fine to use! Normal soap pH is 9-10, so you’re right within range. Good thinking on putting it in the Crock-Pot too! That can help emulsify soap that is starting to separate. 🙂
-Kelsey with Bramble Berry
I have been making cold process soap for a while now and never had any issues with the PH. I have very sensitive skin that would burn with Ivory soap. I always superfat between 5 and 8%. We just moved from a house with well water and never had any reactions with the soap. The house we moved into also has well water but every time we shower, our tub turns blue. When I wash my children in the bathtub their water turns blue as soon as the soap mixes with it!! The stains in the tub are a nuisance as is bathing children in blue water. Haha I did some experiments today just to see if it is the soap which was great for this homeschooling mom to show her children, but causes concern for our water safety. I tested a couple other soaps as well, that I did not make and same reaction. It seems more pronounced in warm/hot water vs. Cold. Anyone else ever have this problem or know what is causing such a reaction between water and soap? Is the water acidic?
Hi Sarah!
I’m not entirely sure why that’s happening! I’m wondering if it’s the well water. I would recommend pH testing the water to see what’s going on. I would also recommend pH testing the soap. Maybe the water and soap are reacting to create that color? If you find it’s a bit acidic, you may want to contact someone to see if you can get it fixed!
-Kelsey with Bramble Berry
pH Test Strips: https://www.brambleberry.com/pH-Strips-1-pack-of-50-P4434.aspx
Thank you Kelsey!
You’re welcome! Let us know how it turns out. 🙂
-Kelsey with Bramble Berry
HI im new to cp soaping so far my lavender ,orange soap test with cabbage turn blue but my beer soap still green after 4 weeks so i did a zap test to all 3 soap no zap what should i do about the beer soap thankyou .
Hi Tammy!
To be on the extra safe side, I would recommend using pH test strips. It sounds like the test results are a bit inconsistent, so the pH strips can give you a definitive pH! You can find those in a hardware store near the hot tub section, or on Bramble Berry: https://www.brambleberry.com/pH-Strips-1-pack-of-50-P4434.aspx
If the soap is around 9-10, it’s just fine to use! If it’s higher than that, it is lye heavy and shouldn’t be used on the skin. It does make great laundry soap though. 🙂
Three DIY Laundry Soap Recipes: https://soapqueen.com/bath-and-body-tutorials/home-crafts/three-diy-laundry-soap-recipes/
-Kelsey with Bramble Berry
Thankyou so much Kelsey
can anyone help me please
You’re welcome! If you have any other questions let me know. 🙂
-Kelsey with Bramble Berry
Just a question about this process in general… If at some point the soap tests 8-10 ph ( say at 4 weeks) does that indicate it is safe? I know that the cure time is for water loss, hardness etc as well as lye dissipation, but could soap that meets the ph safety requirements be used earlier than the 6 weeks?
Hi Marianne!
After the first two days, most of the lye has saponified. We recommend pH testing it after 5 days to be extra safe. After that time, the pH may lower slightly over the next 4-6 weeks, but will likely stay right around 8-10. So, if your soap is around 8-10 after 5 days, it is fine to use!
Technically you can use the soap after those first couple days. If you have a harder recipe (with a high amount of hard oils like coconut and palm), it may be ready to use at 4 weeks! After 4 weeks, you can test one of the bars. If you notice it’s soft or has quite a bit of residue, I would recommend letting the other bars cure another 2 weeks. 🙂
-Kelsey with Bramble Berry
Thanks so much for the clarification, Kelsey!.
You’re welcome Marianne! 🙂
-Kelsey with Bramble Berry
Hello. So great I found this & I sure thank you for posting!!! Since I do not have nor can I afford to order PH strips, this info came from heaven. Not fully understanding what “zap” should feel like, the cabbage method would be my thing. A friend of mine is giving me away her Crock-Pot next week and I’ll be making my first attempt at soap making with the HOT process. The recipe/tutorial I’ll follow is for whipped soap. Can use this method for the PH check? Can it also be used in hot process soap making?
Cheers & thanks!
You’re welcome Marisol, glad you found the post helpful! This method can also be used for hot process soapmaking. I would recommend pulling off a tiny amount of the soap in a heat safe container and dropping a bit of the cabbage mixture on. If it’s a bit too high, you can keep cooking. 🙂
This video has some more great tips on making hot process soap: https://www.youtube.com/watch?v=mxTXwxbpzLM&spfreload=10
-Kelsey with Bramble Berry
I had read this article a while back, but never tried the technique. I made liquid dish soap recently and have it in my pump at my kitchen sink. I pureed blood oranges today- a beautiful red/purple color; I strained the pulp in my nut milk bag. To tie it all together- when I pumped a squirt of soap on the bag, it turned blue! I guess any vegetable or fruit with enough of this flavonoid could work as a pH indicator. I just HAD to share my experience!
Oh that is so cool! The blue was probably a bit surprising at first I imagine. Thanks so much for sharing Cara. I want to give it a try now! 🙂
-Kelsey with Bramble Berry
hello i made today liquid soap from scratch for my liquid shampoo base before diluting i check the ph test with strip the ph is 7 is it neutral , or acetic please do reply me thank you
Hi Sandeep!
A pH of 7 is neutral! Typically, cold process soap has a pH level of 9-10. With a pH of 7, your soap should be nice and gentle. 🙂
-Kelsey with Bramble Berry
question? My soap is not lye heavy. something went haywire with my scale and I wound up with too much oil. That was yesterday. Now, I have cut the batch and it is sooo oily and soft. Pitiful. Expensive. Is there anything that I can do to fix it. And the colors are so pretty, too. Perfect red and spruce green and snow white. I could cry. Any ideas?
Shelby and Thank you
Hi Shelby!
Oh no, I’m sorry about that! I have definitely added a bit too much oil to my batch as well.
Do you know how much extra oil was added to the batch? If it was just a little bit, your soap will be softer but still usable! You can let it cure for 4-6 weeks to help harden the soap. It won’t be as firm as the original recipe, but will still feel great. 🙂
If there is quite a bit of extra oil, you can rebatch the soap with equal or double of a nice firm soap recipe. For instance, our Basic Rebatch has hard oils and is a nice firm recipe: https://www.brambleberry.com/Grated-Rebatch-Soap-Base-Basic-P4540.aspx
The firm rebatch will mix with the softer recipe and help make your soap more balanced. Learn how to rebatch soap in this video: https://soapqueen.com/bath-and-body-tutorials/cold-process-soap/soap-queen-tv-presents-how-to-make-rebatch-soap-2/
-Kelsey with Bramble Berry
what should be the ph level of soap should be for milder soap and shampoo bar ph level
Hi Sandeep!
Typically, soap pH is around 9-10. For a gentler shampoo bar, you can use citric acid to lower than pH level to 7. That makes it a bit more gentle for the hair. 🙂
Learn how to use citric acid in your recipe in the Sudsy Shampoo Bar Tutorial: https://soapqueen.com/bath-and-body-tutorials/cold-process-soap/sudsy-shampoo-bars/
-Kelsey with Bramble Berry
Could you use cabbage water in soap as a natural colorant? Does the color fade?
Hi Erica!
Interesting thought! We haven’t actually experimented with this. Many natural colors tend to fade or morph in cold process soap, due to the harsh pH environment. You could certainly give it a try, although I’m not quite sure what kind of color you’d end up with…so you’ll want to be open minded with the results :). From a quick google search, it sounds like it gives soap a blue color. I hope this helps!
-Amanda with Bramble Berry
Hello !! I have been doing the zap test with all of my bars of soap to check if they are not lye heavy. So far, none of my soaps have zapped me… But just to make sure I’m on the safe side, I purchased a bottle of phenolphtalein in amazon… the thing is, it’s not so clear to me how it works !! any help will be very much appreciated !!
Hi Rosy!
To use phenolphtalein, place a drop directly onto the soap. Then, check the color against the guide provided. Also, make sure to read the directions, as certain brands may have different instructions. 🙂
-Kelsey with Bramble Berry
dear Kelsey,
The supplier didn’t send a guide, nor are there any instructions on the bottle… that’s why I’m so confused ! I’ve read on other websites that the phenolphtalein should be clear after placing a drop on the soap (with distilled water), but mine turn out pink and the soap doesn’t zap, so I don’t know what to make of that !!
Hi Rosy!
From what I understand, clear means the pH is below 7, light pink means the pH is 7 and dark pink means the pH is above 7. Typically, soap pH is 9-10, so it should be dark pink.
You may like our pH strips! They measure a pH range of 1-14, and include a handy color chart. 🙂
pH Strips: https://www.brambleberry.com/pH-Strips-1-pack-of-50-P4434.aspx
-Kelsey with Bramble Berry
thank you Kelsey ! will definitely purchase your ph papers next time I place an order.
You’re welcome Rosy! 🙂
Rosy – Same here – the supplier never sent instructions or a guide with my phenolphtalein! What’s UP with that? And I’ve always felt that the “zap test” was vague at best, so I’ve just made a habit of sampling soap from each batch (which tends to accumulate bars in our shower, lol). Thanks, Kelsey for clearing this up.
You’re welcome Shaman! 🙂
Hi,
Why are other natural/homemade soaps such as Nubian Heritage and COPA soap consistently test at 8? Is it possible to add a little lemon juice to make it a little more mild then 9 or 10? I live near Lancaster PA and saw an Amish woman use lemon juice to bring down PH. Is this something homemade Soap’ers can try?
Thanks
Tracey
Hi Tracey!
While I can’t speak for other soapers, I can tell you the soaps we make are usually around 9-10.
If you want your pH level a little lower, you can use citric acid! To use it, combine .5 ounces of citric acid with with 4 tablespoons of distilled water. You can add that mixture at trace. Keep in mind it will accelerate, so you’ll need to work a little faster. 🙂
We used citric acid in our Sudsy Shampoo Bars with great results: https://soapqueen.com/bath-and-body-tutorials/cold-process-soap/sudsy-shampoo-bars/
-Kelsey with Bramble Berry
Hi I made a soap today and I forgot to add 100 grm of my rice bran oil. superfat was 10% and lye 133 grm AND WATER 380, but I add only 130 grm of lye and one table spoon of glycerin. do you think my soap is heavy lye now. I try this technique but I think i panic and I really cant recognize the color. what shall I do?
Shall I re-batch my soap in crock pot and I add 100 grms of rice bran oil? or soap will be fine after WEEKS???
Please someone advise me,
Thanks
Hi Shahrzad!
I would recommend running that recipe through the Lye Calculator again without the rice bran oil. That will let you know if the soap is potentially lye heavy!
Lye Calculator: https://www.brambleberry.com/Pages/Lye-Calculator.aspx
If your soap is lye heavy, you can still use it! You can get the soap to the right pH level with the Hot Process Hero method: https://soapqueen.com/bath-and-body-tutorials/tips-and-tricks/hot-process-hero-2/
-Kelsey with Bramble Berry
Hi , yesterday I made Cp Soap, I tried red cabbage technique today when I removed my soap from the mold. Red Cabbage juice turn to green!!!! Shall i try same test again? it means after 2 weeks….did I destroy my soap???
Hi Shahrzad!
Most of the saponification, or soapmaking process, happens within the first 48 hours. That means the first two days after you make your soap, the pH level may still be a bit high. I would recommend testing your soap again in a couple of days, and again at a week or so. By that time, the pH level should be around 10. 🙂
If not, don’t worry! You can turn that soap into laundry soap. Check out how in the Three DIY Laundry Soap Recipes post: https://soapqueen.com/bath-and-body-tutorials/home-crafts/three-diy-laundry-soap-recipes/
-Kelsey with Bramble Berry
hmmm……so would red cabbage be a good natural colorant to make blue soap?
dehydrate it and grind it.
does it changes from green to blue when the lye is done?
or would it stay green?
nevermind, i googled it and people who tried it said it made the soap really stinky.
Hi jnl!
We tested cabbage juice as a colorant for soap with not so great results. The cabbage juice changed colors as the soap set up. In the end, it was a pastel pink color, but that faded out in a week or so. It does also have a pretty strong scent. Other natural colorants like natural indigo powder may be better for a blue soap. 🙂
-Kelsey with Bramble Berry
Natural indigo powder: https://www.brambleberry.com/Natural-Indigo-Powder-P5727.aspx
I’m confused by the pH scale you posted. On your scale yellow is a pH of 5, which would be acidic. But the lye heavy soap turned yellow. Also wouldn’t neutral just stay purple?
Hi Hayley!
Our pH scale is an illustration to show an example of the different levels. The colors don’t coordinate with the cabbage test. Sorry for any confusion! 🙂
-Kelsey with Bramble Berry
Awesome idea! Does it also work with creamsoap? (Not that I have much succes with that one, but we keep hoping :p)
Btw, this really reminds me of a halloween drink!
http://www.lowcarbsosimple.com/witches-brew-color-changing-halloween-drink/
Hi Kim!
We haven’t tried this test with cream soap, but I believe it would work! If you give it a try, let us know how to goes. 🙂
Also, thanks so much for sharing that link! The drink looks like a lot of fun to make.
-Kelsey with Bramble Berry
This is really cool! I knew about the zap test and pH strips, but this is really fun! I may have to try this with my kids. Which means I will have to make laundry soap so they can compare it to my regular soap bars. Fun!
Hi Margaret!
This would be such a fun project with your kids! It’s a blast watching the colors change. 🙂
-Kelsey with Bramble Berry
I have to say no to zap testing. There’s no reason not to be using phenolphthalein of PH strips today as you can get them virtually anywhere and they’re dirt cheap. Besides being dangerous, there’s no way you can ascertain a soap PH level with a tongue test. If you must work without either of those – or the cabbage juice – then just rub a little bit of the wet soap on a tiny section of skin instead. If you’re going to get a slight lye burn it’s better that it’s on the back of your hand than your tongue. That aside, the cabbage juice trick is neato keen. That’s handy to know.
Hi Cutter!
In our tests and recipes, we’ve found that the zap test is an effective method for testing. We do definitely recommend checking the soap to make sure there are no lye pockets. If not, that test is perfectly safe. It’s simply a quick tap on your tongue!
However, there are plenty of other methods out there if you aren’t comfortable with the zap test! pH strips and this cabbage test work great. 🙂
-Kelsey with Bramble Berry
That is so COOL!
Hi Molly!
We thought so too! We had a lot of fun testing out the soap in our lab. Glad you enjoyed the post. 🙂
-Kelsey with Bramble Berry
Wonderful article. It was very informative and I will try this method. I am currently using pH test strips which can be quite expensive.
Hi Jocelyn!
So glad you like the post Jocelyn! It’s great to have a couple of ways to test for lye heaviness. 🙂
-Kelsey with Bramble Berry
I’ve done it this way for a long time, and just wanted to give you a little hint, after you’ve made your cabbage water, freeze it in ice cube trays, because you only need a few drops for each batch at a time.
Hi Colleen!
That’s such an awesome tip, thanks so much for sharing! What a great way to reuse the extra juice. 🙂
-Kelsey with Bramble Berry
Thank you for posting that! It was going to be my question, if I could freeze it and it would still work. Now I know.
That’s a great tip Colleen! How long could you keep it in the freezer for it to still work if you only make soap periodically?
This is great! I’ve only made 4 batches of CP soap and every time I’m paranoid about the numbers. This will be very useful the next time I make a batch!
Hello!
That’s awesome, glad you found the article helpful! Have fun giving this technique a try. 🙂
-Kelsey with Bramble Berry
Oh! This explains why, when I was rebatching [very brittle] soap, and added raspberry tea steeped in distilled H2O, the deep ruby red tea instantly turned green upon contact with the soap. My soap was lye heavy! I must read more about flavins!
Thanks! Very interesting!
Hi Penny!
Oh that’s really interesting! It sounds like that tea was reacting to the lye. Glad you like the article Penny. 🙂
-Kelsey with Bramble Berry
Wow, who would of thunk?!? I wanna know who thought of this? lol But seriously thank you for the information! It’s always nice to have another way to check incase one is borderline.
Hi Cathy!
I thought it was so interesting that you can test pH with cabbage! Glad you liked the post. 🙂
-Kelsey with Bramble Berry
What do you do with the soap after testing? Do you just rinse it off and dry it, and then you can sell it? Or is that the one you keep for yourself?
Hi Martina!
The cabbage juice rinses off with a little bit of 99% isopropyl alcohol, so you can definitely still sell those bars! Leaving the juice on there for hours may stain the soap, but otherwise it will wipe away clean. 🙂
99% isopropyl alcohol: https://www.brambleberry.com/Isopropyl-Alcohol-99-16-oz-P5682.aspx
-Kelsey with Bramble Berry
This is awesome, Anne-Marie!! I am taking Chemistry this year, and am right now studying acids and bases, so this post was extremely timely! I just did an experiment a few days ago that used red cabbage water to test the pH of a solution. Chemistry has definitely deepened my understanding and appreciation of soap making!
I have always wondered if you could color soap with cabbage water and if the soap would change colors as the soap cured. I’m not sure if the color would make it through the process or not, but it might be worth a try. Anyhow, this pH test is so neat! I want to try it! And since I still have some red cabbage left over from my experiment…
Hi Olivia!
That’s great, what perfect timing! There is definitely a lot of science in soapmaking. 🙂
Also, we have tried coloring soap with cabbage juice with not so great results. Because of all the pH changes during the soapmaking process, the soap changes colors quite a bit. The final soap turned a pastel pink color, but faded out after a couple weeks.
Although the final color isn’t very bright, the process is very fun to watch! It may be nice for a small test batch just so you can watch it change colors. 🙂
-Kelsey with Bramble Berry
That would be very neat to try!